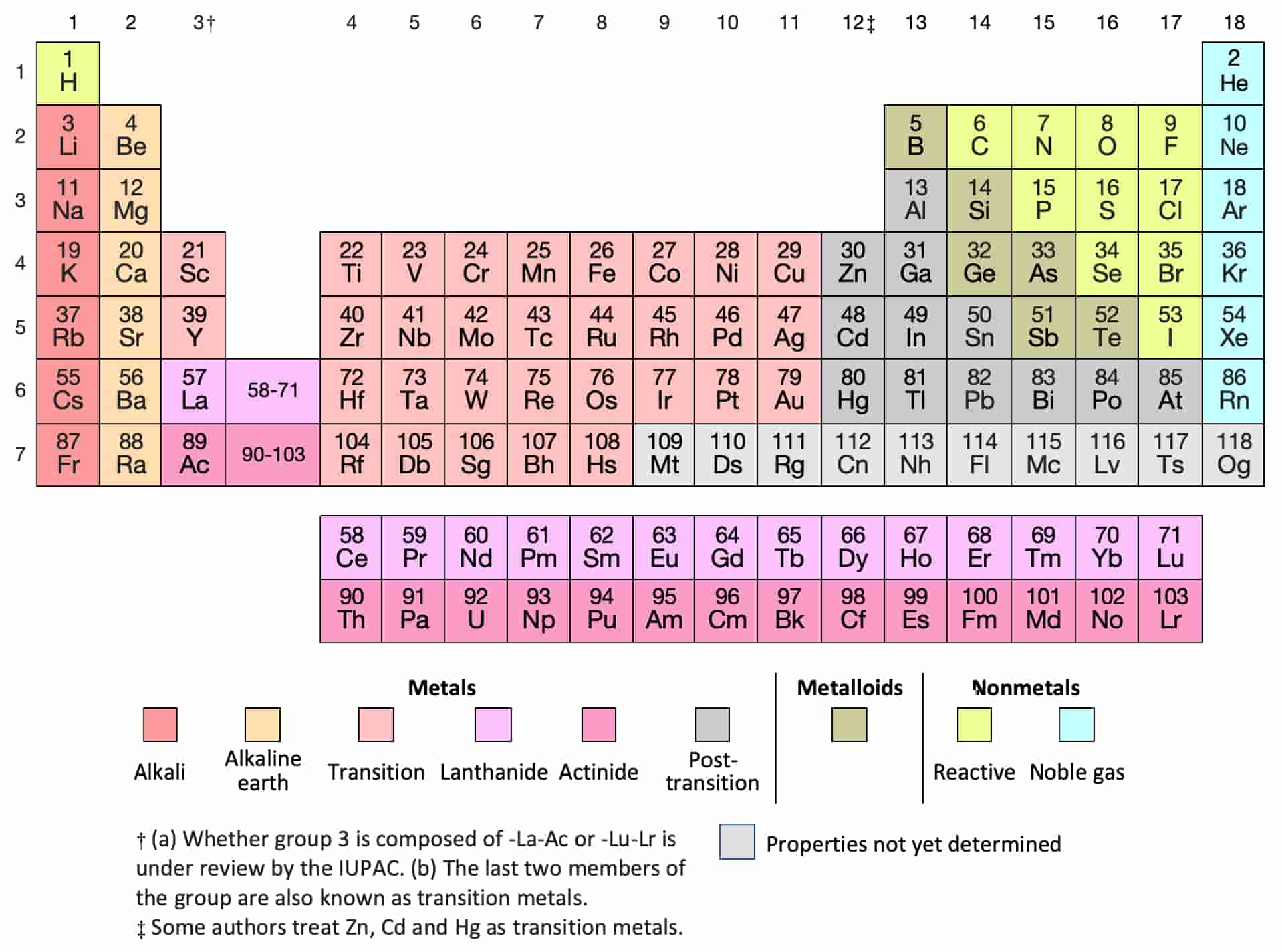
In accord with the octet rule -and to become more energetically stable - atoms gain, lose, or share valence electrons in an effort to obtain a noble gas configuration in their outer shell. The electrons in the highest energy level are called valence electrons. The electrons in an atom are located at different energy levels. For instance, a unimetal will have a valence of 1. For example, uni/mono stands for 1, while bi/di stands for 2, tri for 3, and tetra for 4. Oftentimes, Greek prefixes are used to describe an atom ’s or a materials ’ valency.
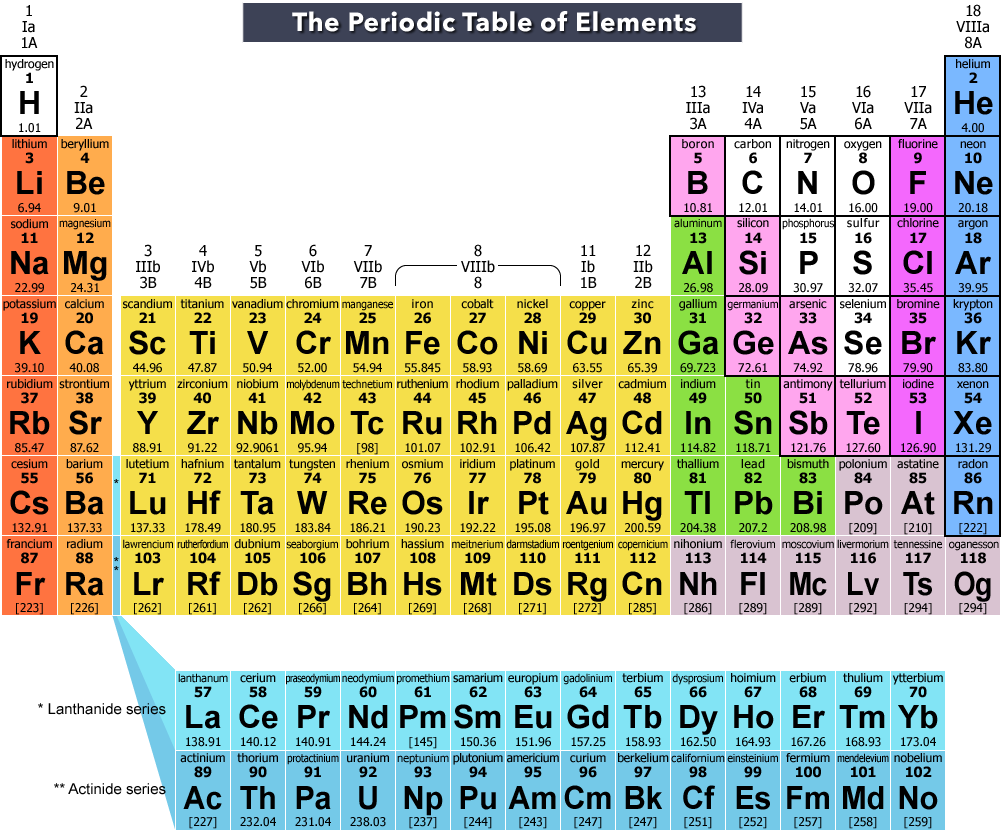
The concept was formulated in the nineteenth century as a way to organize formulas of the various chemical compounds. The term valence, also referred to as valency or valence number, is derived from the Latin word for strength and can reflect an element ’s strength or affinity for certain types of reactions. Thus, the value of valence is associated with the number of electrons, if any, that an element loses or accepts from another atom during a chemical reaction. It also refers to the type of reactions the element will undergo. It is important to recognize which numbering system is being used and to be able to find the number of valence electrons in the main block elements regardless of which numbering system is being used.Valence, in chemistry, refers to a number assigned to elements that reflects their ability, or capacity, to react (combine) with other elements. You will come across periodic tables with both numbering systems. In this numbering system, group 1A is group 1 group 2A is group 2 the halogens (7A) are group 17 and the noble gases (8A) are group 18. The elements in this group are also gases at room temperature.Īn alternate numbering system numbers all of the \(s\), \(p\), and \(d\) block elements from 1-18. We will learn the reason for this later, when we discuss how compounds form. These elements also have similar properties to each other, the most significant property being that they are extremely unreactive, rarely forming compounds. This group contains very reactive nonmetal elements. Group 7A (or 17) elements are also called halogens. It is important to recognize a couple of other important groups on the periodic table by their group name.

Remember, Mendeleev arranged the table so that elements with the most similar properties were in the same group on the periodic table. The same pattern is true of other groups on the periodic table. Once again, because of their similarities in electron configurations, these elements have similar properties to each other. Group 2A is also called the alkaline earth metals. Although most metals tend to be very hard, these metals are actually soft and can be easily cut. Group 1A is also known as the alkali metals. Because of their similarities in their chemical properties, Mendeleev put these elements into the same group. The elements in 1A are all very reactive and form compounds in the same ratios with similar properties with other elements. This is what causes these elements to react in the same ways as the other members of the family. All of the 1A elements have one valence electron. A group is a vertical column of the periodic table. Remember that Mendeleev arranged the periodic table so that elements with the most similar properties were placed in the same group.
